Why Almost Every U.S. Hospital Is Building Safety Stock of Critical Pandemic Medications

According to a new survey of Premier members, 88 percent of hospitals and health systems are building safety stocks of critical medications to prepare for a caseload surge of COVID-19 throughout the summer and fall.
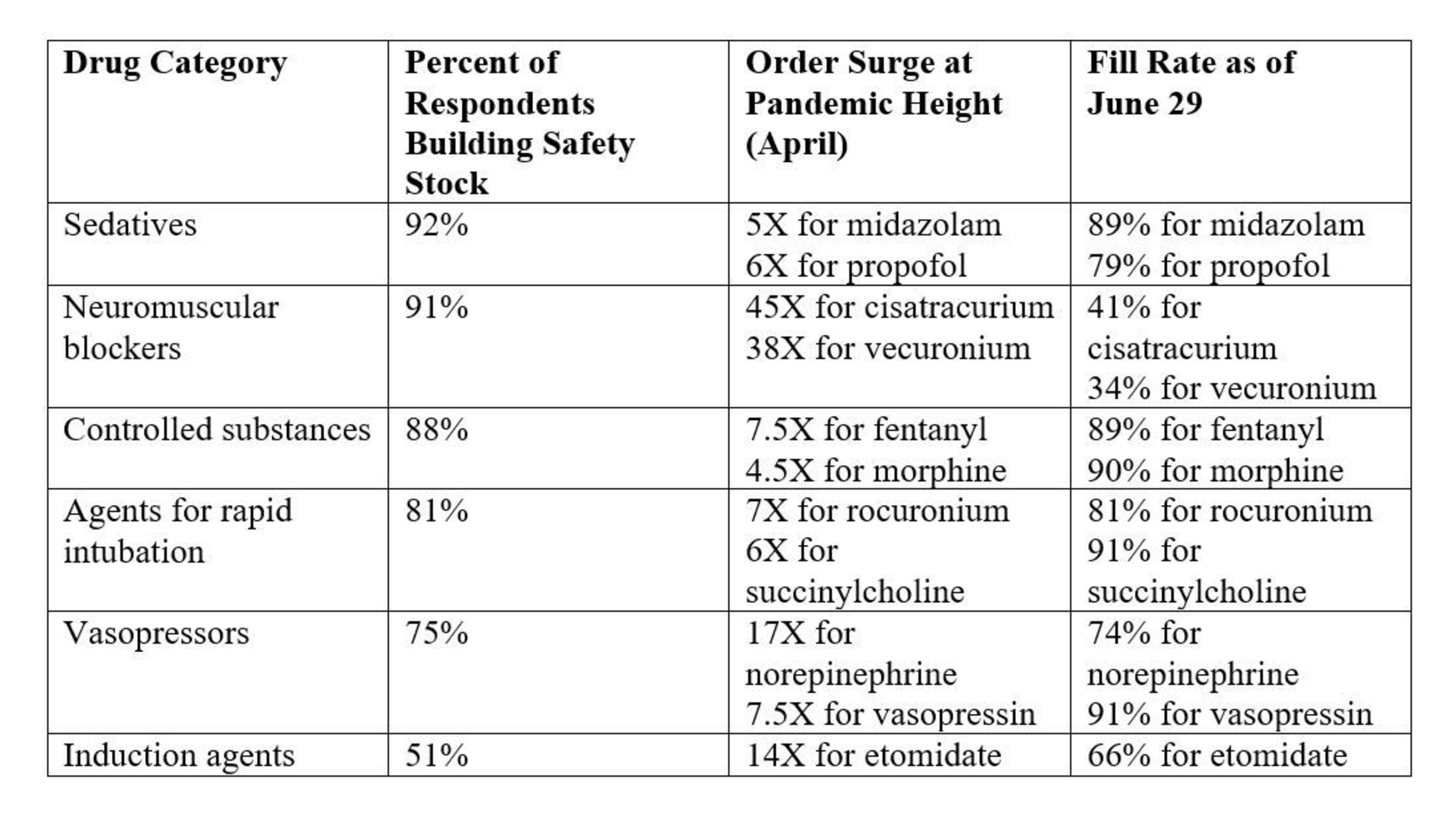
A majority of survey respondents (51 percent) reported building safety stocks of at least one month, and another 25 percent said they were planning to stock at least a two-month supply. The most commonly cited COVID-19 drugs for inclusion in a hospital’s safety stock were sedatives, followed by neuromuscular blockers and controlled substances.
Additionally, many hospitals and health systems are also building safety stocks for drugs used to treat COVID-19 co-morbid conditions and associated complications, such as respiratory inhalers to manage asthma (cited by 64 percent of respondents), emergency syringes for cardiac care (53 percent), antibiotics for secondary infections (51 percent) and IV fluids to manage dehydration (49 percent).
Nearly three-quarters (72 percent) of respondents say they will pay for added safety stock out of their existing pharmacy budgets, while 28 percent say additional funds will be allocated to manage the expense.
Why Build Safety Stock?
While the Premier survey did not expressly ask why hospitals are building safety stock, early experiences accessing stockpiles this spring highlighted problems associated with relying on the government for product replenishment. Not only did the Strategic National Stockpile (SNS) run out of products in early April, but the federal government has repeatedly stated any products it does have are not intended for use by states or individual hospitals in those states. The message: Hospitals are on their own and any stockpiles they access must be homegrown.
To boot, Premier reported last week that nearly 90 percent of healthcare providers are contributing to stockpiles of critical medical supplies and drugs, as directed by either the state or health system leadership.
In addition, while there were no widespread, long-term shortages for most drugs needed to manage COVID-19 patients this spring, many hospital fear that was just a stroke of luck.
For years, the industry has been dealing with pervasive drug shortages for a number of products, including those needed for COVID-19 patient care. Although most manufacturers have prepared and have months of finished dose and active ingredient supplies available today, data shows that when cases surge, ordering for certain drugs can increase by as much as 4,000 percent. At that rate, months of supply on hand quickly turns to no supply, and without safety stock, hospitals could be caught flat-footed.
What’s Next?
Since the pandemic began, Premier has been monitoring national ordering and fill rates for more than 250 medications that have been identified as critical to providing emergency and COVID-19 care.
In some cases, we’ve been able to secure a dedicated supply of the medication in question by signing long-term, guaranteed purchase agreements with the manufacturer through the ProvideGx generics sourcing program – a program that has been able to meet all ordering requests from our members, despite demand surges of more than 150%.
In other cases, we’ve been able to change distributor allocations to route products to hotspots that have the greatest need, secure additional supply from the manufacturers or arrange for direct emergency shipments in cases of dire need. We’ve also shared the data with the U.S. Food and Drug Administration (and continue to do so weekly) to provide advance notice of potential shortages and inform policies that could avert shortages.
Looking ahead, Premier continues to advocate for major reforms to the SNS making it easier to access and ensuring that future supply efforts are part of a coordinated, methodical, national effort. For instance, Premier is pushing for the SNS to move away from a central repository to a coordinated hub-and-spoke model made up of federal resources linked to state and local resources, with an emphasis on collaboration between all those maintaining inventories.
Further reading: Premier’s supply chain reflections and recommendations on preparing for the next surge or pandemic.
Survey Methodology
Premier’s survey was conducted from June 11-June 29, 2020, and sent to a representative portion of acute care providers in the Premier membership. Respondents included nearly 90 health systems from across the country.
Wayne is responsible for the contracting relationship with the pharmaceutical industry, including manufacturers, distributors and wholesalers, pharmacy technology and services, plasma derived products, vaccines and biosimilars.
Mike helps Premier's member health system pharmacy leaders practice high-quality care at the lowest possible cost, including integration of pharmacy services across the care continuum that translates into sustainable financial performance and improved patient care outcomes.
Article Information
Wayne is responsible for the contracting relationship with the pharmaceutical industry, including manufacturers, distributors and wholesalers, pharmacy technology and services, plasma derived products, vaccines and biosimilars.
Mike helps Premier's member health system pharmacy leaders practice high-quality care at the lowest possible cost, including integration of pharmacy services across the care continuum that translates into sustainable financial performance and improved patient care outcomes.